- Home
- About
- Contact
- Civilization revolution 2 xbox one
- Final draft 9 manual
- Pier park pcb
- Plagiarism checker for code
- Proteus for android
- Silver surfer vaporizer
- What men want 123 movies
- Terrarium tv app for android
- Free download real football 2012 for android
- Chinkee tan network marketing
- Rhino cad cam
- Marvel ultimate alliance pc multiplayer
- Nonpolar hydrophobic amino acids
- Charly mcclain fly into love


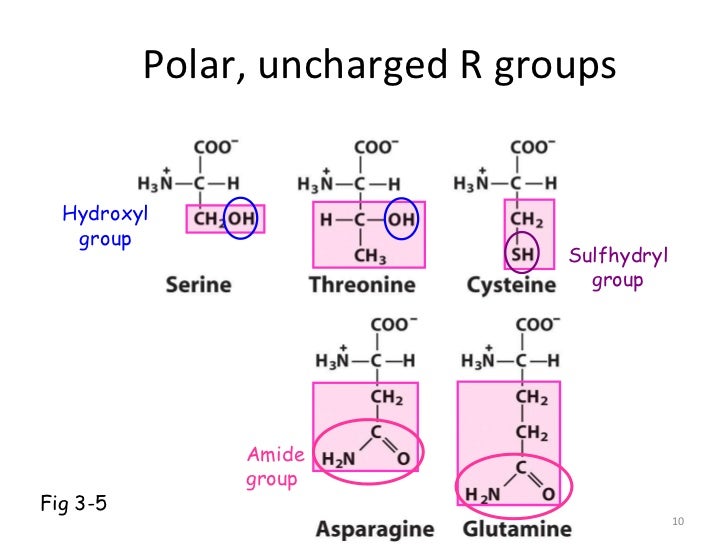
Disulfide bonds play a special role in the structures of many proteins by forming covalent links between parts of a protein molecule or between two different polypeptide chains. The disulfide-linked residues are strongly hydrophobic (nonpolar). Cysteine is readily oxidized to form a covalently linked dimeric amino acid called cystine, in which two cysteine molecules or residues are joined by a disulfide bond (Fig. Asparagine and glutamine are the amides of two other amino acids also found in proteins, aspartate and glutamate, respectively, to which asparagine and glutamine are easily hydrolyzed by acid or base. The polarity of serine and threonine is contributed by their hydroxyl groups that of cysteine by its sulfhydryl group and that of asparagine and glutamine by their amide groups. This class of amino acids includes serine, threonine, cysteine, asparagine, and glutamine. Polar, Uncharged R Groups The R groups of these amino acids are more soluble in water, or more hydrophilic, than those of the nonpolar amino acids, because they contain functional groups that form hydrogen bonds with water.
This accounts for the characteristic strong absorbance of light by most proteins at a wavelength of 280 nm, a property exploited by researchers in the characterization of proteins. Tryptophan and tyrosine, and to a much lesser extent phenylalanine, absorb ultraviolet light. Tyrosine and tryptophan are significantly more polar than phenylalanine, because of the tyrosine hydroxyl group and the nitrogen of the tryptophan indole ring. The hydroxyl group of tyrosine can form hydrogen bonds, and it is an important functional group in some enzymes. All can participate in hydrophobic interactions.
Aromatic R Groups Phenylalanine, tyrosine, and tryptophan, with their aromatic side chains, are relatively nonpolar (hydrophobic). The secondary amino (imino) group of proline residues is held in a rigid conformation that reduces the structural flexibility of polypeptide regions containing proline. Although the R group of histidine is shown uncharged, its p Ka (see Table 1.1) is such that a small but significant fraction of these groups are positively charged at pH 7.0. The unshaded portions are those common to all the amino acids the portions shaded in red are the R groups. The structural formulas show the state of ionization that would predominate at pH 7.0. TABLE 1.1 Properties and Conventions Associated with the Common Amino Acids Found in ProteinsįIGURE 1.1 The 20 common amino acids of proteins. Proline has an aliphatic side chain with a distinctive cyclic structure. Methionine, one of the two sulfur-containing amino acids, has a nonpolar thioether group in its side chain. Although it is formally nonpolar, its very small side chain makes no real contribution to hydrophobic interactions. The side chains of alanine, valine, leucine, and isoleucine tend to cluster together within proteins, stabilizing protein structure by means of hydrophobic interactions. Nonpolar, Aliphatic R Groups The R groups in this class of amino acids are nonpolar and hydrophobic. Within each class there are gradations of polarity, size, and shape of the R groups. The structures of the 20 common amino acids are shown in Figure 1.1, and some of their properties are listed in Table 1.1. The polarity of the R groups varies widely, from nonpolar and hydrophobic (water-insoluble) to highly polar and hydrophilic (water-soluble). The topic can be simplified by grouping the amino acids into five main classes based on the properties of their R groups (Table 1.1), in particular, their polarity, or tendency to interact with water at biological pH (near pH 7.0). Knowledge of the chemical properties of the common amino acids is central to an understanding of biochemistry.
